Exploring the Properties of Enzymes: An In-Depth Look
Table of Contents
ToggleProperties of Enzymes (Physical, Chemical, and General Properties)
Enzymes are natural catalysts made by living cells. They speed up chemical reactions in the body without being used up. Enzymes help digest food, support energy production, and build important molecules like DNA and proteins.
This guide will describe the important properties of enzymes in a clear, simple way. You will learn physical traits (how enzymes behave as substances), chemical traits (what they are made of), and the general properties of enzyme action (how they work during reactions).
Properties of enzymes
Here are the important properties of enzymes most students and readers focus on:
- They have high catalytic power, so small amounts can speed up reactions.
- They show specificity, meaning they act on specific substrates.
- They work best at an optimum temperature and optimum pH.
- Many enzyme reactions are reversible, depending on conditions.
- Many enzymes need helpers called cofactors or coenzymes.
- They show saturation, where the reaction rate rises and then levels off.
- They can be controlled by inhibitors, activators, and other cell systems.
To understand the main enzyme properties, it helps to group them into:
- Physical properties
- Chemical properties
- General working properties
1) Physical properties of enzymes
The physical properties of enzymes affect how stable they are and how they behave in water.
Key physical points
- High molecular weight: Most enzymes are large biomolecules, usually proteins.
- Colloidal nature: In water, many enzymes behave like colloids. They disperse well but do not act like small salt molecules.
- Heat sensitivity (thermolabile): High heat can change an enzyme’s shape and reduce activity. This shape change is called denaturation.
- Optimum temperature: Many human enzymes work best near body temperature.
- Stability in dry form: Enzymes are often more stable when dry than when stored in solution.
2) Chemical nature of enzymes
Most enzymes are proteins, but many need a non-protein helper to work. This is a major part of the properties of enzyme structure and function.
Key terms
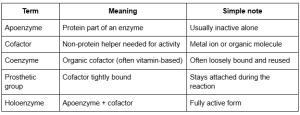
Common cofactors
- Metal ions: Mg²⁺, Zn²⁺, Fe²⁺/Fe³⁺
- Coenzymes: NAD⁺, FAD (often vitamin-related)
Why this matters: Without the right helper, the enzyme may not bind the substrate well or may not complete the reaction. This is one of the most tested enzyme properties in biochemistry.
3) General properties of enzymes
This section explains the general properties of enzyme action during reactions.
A) Catalytic power
Enzymes speed up reactions by lowering activation energy. As a result, reactions that would be too slow can happen quickly at normal temperatures.
A useful idea here is the turnover number (kcat). It shows how many substrate molecules one enzyme molecule can convert per second under ideal conditions. It is a simple way to understand how powerful enzymes can be.
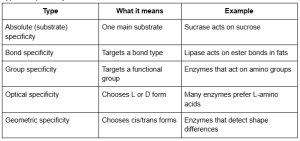
B) Specificity
Specificity means an enzyme’s active site fits certain substrates better than others. This selectivity helps prevent unwanted reactions and keeps body processes under control. Specificity is one of the most important properties of enzymes.
Types of specificity
C) Are enzyme-catalyzed reactions reversible?
Many enzyme reactions can run forward or backward. The direction depends on:
- The concentration of reactants and products
- Energy conditions inside the cell
- What the pathway needs at that time
Some steps appear one-way in cells because the product is removed quickly, or because conditions strongly favor one direction.
D) Effect of temperature and pH on enzyme activity
Temperature and pH can change an enzyme’s shape and the charge of its active site.
Temperature
- Low temperature slows reactions because molecules move less.
- High temperature can denature enzymes, so activity drops sharply.
pH
- A change in pH can change active-site charge and shape. If the fit changes, the substrate binds poorly.
Simple examples
- Salivary amylase works best near neutral pH (mouth).
- Pepsin works best at acidic pH (stomach).
These are classic properties of enzyme behavior seen in labs.
4) Enzyme kinetics
Enzyme kinetics explains how reaction speed changes under different conditions. It is often included when discussing properties of enzymes in experiments.
Substrate concentration and saturation
- As substrate concentration increases, the reaction rate increases.
- After a point, all active sites are occupied.
- The rate then levels off (saturation).
Two common terms
- Vmax: the fastest rate when all enzyme active sites are filled.
- Km: the substrate level where the reaction rate is about half of Vmax. Lower Km often means the enzyme works well at lower substrate levels.
5) Enzyme regulation
Cells regulate enzymes so reactions stay balanced. Regulation is one of the most practical enzyme properties to understand.
Main regulation methods
- Competitive inhibition: inhibitor competes with the substrate for the active site.
- Noncompetitive inhibition: inhibitor binds elsewhere and reduces activity even with substrate present.
- Allosteric regulation: a molecule binds at a control site and turns activity up or down.
- Feedback inhibition: the final product slows an earlier enzyme to prevent overproduction.
- Gene-level control: cells make more or less enzyme depending on need.
6) Industrial applications of enzymes
Enzymes are useful in industry because they are efficient and selective. Many industries choose enzymes based on the important properties of enzymes, especially stability and specificity.
Detergents
- Proteases remove protein stains.
- Lipases break down grease.
- Limits: high heat or wrong pH reduces activity.
Food and beverages
- Amylase breaks starch into sugars (baking and brewing).
- Lactase supports lactose-free dairy.
- Limits: conditions must be controlled for taste and texture.
Textiles
- Cellulase helps with fabric finishing.
- Limits: overuse can weaken fabric.
Pharmaceuticals (brief)
- Some therapies use enzymes, such as enzyme replacement therapy.
- Limits: strict purity and safety testing are required.
FAQ
What are the main properties of enzymes?
The main properties of enzymes include catalytic power, specificity, sensitivity to temperature and pH, reversibility in many reactions, and the need for cofactors in many cases.
What is the difference between apoenzyme and holoenzyme?
An apoenzyme is the protein part alone. A holoenzyme is the active form, made of the apoenzyme plus its cofactor.
Why do enzymes stop working at high temperature?
High heat can denature an enzyme. Denaturation changes the active site shape, so the substrate no longer fits well.
Do enzymes get used up in reactions?
No. Enzymes are not consumed. They can be reused unless damaged by heat, extreme pH, or harsh chemicals.
Conclusion
The properties of enzymes include physical stability, chemical structure (often with cofactors), and reaction behavior such as specificity, reversibility, and regulation. Once you understand these enzyme properties, it becomes easier to explain digestion, metabolism, lab experiments, and industrial enzyme use.

